First year organic chemistry at Bristol
Course details
Course lecturers:
Dr Paul Wyatt (PJW)
Professor Varinder Aggarwal (VKA)
Professor Kevin Booker-Milburn (KIBM)
Recommended textbooks: see my list
Date: October 2007—May 2008.
See also: Wikipedia notes on first year organic chemistry.
Table of contents
- Structure and Reactivity (PJW)
- Useful links
- Wikipedia notes
- Terminology
- Reactions and Synthesis with Carbonyl Compounds (VKA)
- Reactions
- Reductions
- The Chemistry of Carbon-Carbon Double Bonds (KIBM)
- Alkenes
- Reactions of alkenes
- Electrophilic addition in general
- Hydrohalogenation
- Hydration
- Hydroboration
- Bromination
- Reduction
- Oxidation
- Alkynes
- Aromatic chemistry
I've compiled a list of Wikipedia articles that cover the topics in this lecture course on my Wikipedia user page: http://en.wikipedia.org/wiki/User:Benjah-bmm27/degree/1/PJW.
Much of the terminology used in this course and throughout chemistry (such as electrophile, nucleophile, inductive effects, SN2) came from the late Sir Christopher Kelk Ingold (UCL biography, Wikipedia article), one of the founders of physical organic chemistry.
Reduction of carbonyls to alcohols with NaBH4.
Jones oxidation with CrO3.
My working for the first KIBM tutorial: download PDF (1.70 MB). Caution — not necessarily 100% correct (although mostly right).
Wikipedia articles: Electrophilic addition, Markovnikov's rule, Syn and anti addition
Electrophilic addition in general: March p. 999.
Wikipedia article: Hydrohalogenation
Hydrohalogenation AKA hydro-halo-addition. March pp. 1029–1031.
Wikipedia article: Hydration reaction, Oxymercuration reaction
Hydration AKA hydro-hydroxy-addition. March pp. 1032–1035
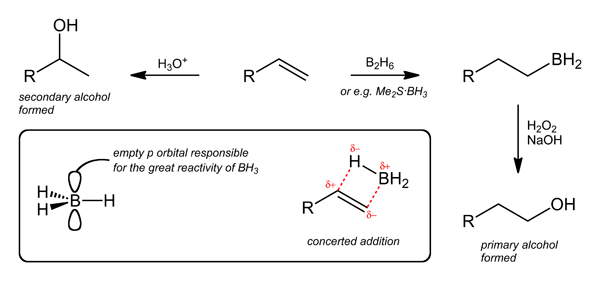
Wikipedia article: Hydroboration-oxidation reaction
Hydroboration, March pp. 1075-1082.
Wikipedia articles: Halogen addition reaction, Halonium ion
Bromination AKA dibromo-addition, March pp. 1148-1152.
Wikipedia articles: Hydrogenation, Palladium on carbon
Hydrogenation AKA dihydro-addition, March pp. 1053-1066
Reduction with H2(g) and a Pd/C catalyst.
Wikipedia articles: Alkene#Oxidation, Epoxide, meta-Chloroperoxybenzoic acid
Epoxidation AKA epi-oxy-addition, March pp. 1169-1179
Epoxidation of an alkene with a peracid is known as the Prilezhaev reaction and its mechanism is sometimes called the Butterfly Mechanism.
Wikipedia articles: Osmium tetroxide#Oxidation of alkenes, Upjohn dihydroxylation
Dihydroxylation AKA dihydroxy-addition, March pp. 1162-1168
Wikipedia articles: Ozonolysis, Molozonide, Ozonide, 1,3-Dipolar cycloaddition
Ozonolysis, March pp. 1736-1742
- Reduce alkynes (Lindlar for Z, sodium in liquid ammonia for E)
- Wittig carbonyls
- Eliminate HX: E1 vs. E2 mechanisms
